You may want to go to a specific submission workspace (webpage) to view or update its contents, submit it for review, review it, or take other actions. Note that your access to a submission is personalized based on your role in the system and the role you play in relation to the particular submission. In addition, the actions you can take on a submission are personalized.
-
To view a submission workspace (webpage), click the submission name when you find it in a list of studies.
-
To find a list that includes the submission name, click:
-
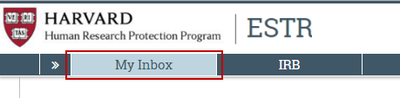
My Inbox (only items that require attention): Click the My Inbox link in the top navigation header. This list displays submissions assigned to you for action, such as submissions you are preparing to submit or submit response to a requested clarification.
-OR- -
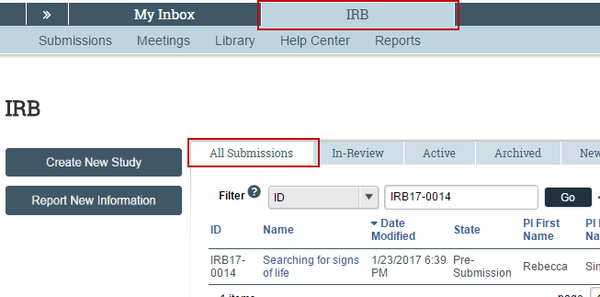
IRB (all items to which you have access): Click IRB in the navigation header and select the All Submissions tab. The lists display workspaces for all studies, continuing reviews, modifications, and reportable new information (RNI) entered into the system that you have permissions to view.
-
My Inbox (only items that require attention): Click the My Inbox link in the top navigation header. This list displays submissions assigned to you for action, such as submissions you are preparing to submit or submit response to a requested clarification.
| TIPS |
|
Managing Submission Permissions
Permissions on a submission are different, depending on your role on the study. The Principal Investigator, study team and guest permissions are different than IRB office staff, and ancillary and IRB reviewers. Principal Investigators, study team members and guests have the following permissions:
|
ESTR Role* |
Principal Investigator |
Primary Contact |
PI Proxy |
Study Team Member |
Guest |
|
Receives ESTR system notifications |
✔ |
✔ |
✔ |
||
|
Can create submissions on behalf of the PI |
✔ |
✔ |
✔ |
✔ |
|
|
Can submit initial submissions |
✔ |
|
|
|
|
|
Can submit follow-on submissions on behalf of the PI Modification/Update, Continuing Review, and Closure only. |
✔ |
✔ |
|||
|
Can submit RNI submissions on behalf of the PI RNI can only be submitted by the person who created the RNI in ESTR. |
✔ |
✔ |
✔ |
✔ |
|
|
Can complete the Copy Submission activity |
✔ |
. |
|
||
|
Modification required to add or update this role |
✔ |
✔ |
✔ |
||
|
Has approval to conduct human subjects research activities/is listed on the personnel roster |
✔ |
✔ |
✔ |
||
|
Has view-only access to ESTR records** |
✔ |
*To manage permissions, see sections for how to change a primary contact, assign proxies, change study team members, copying a study, and add guests.
** For view access to many records in a department, school, or under the purview of an IRB, contact estrhelp@harvard.edu for expanded guest access.
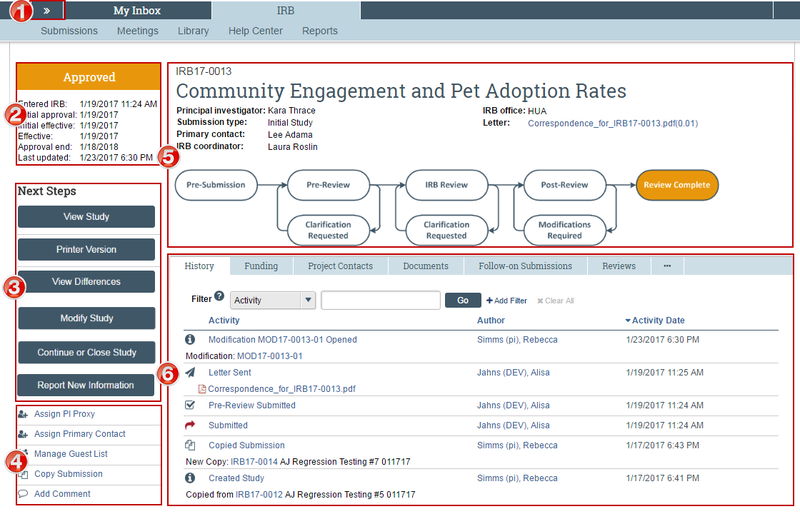
Submission Workspace Overview
nitial/Main-study, Continuing Review, Modification/Update and Reportable New Information submission workspaces are formatted similarly. However, the following general concepts apply to navigation:
- The Initial/Main study workspace (labeled with numbers IRB##-#### or #####) always has the most current approved information and materials for a study.
- The Site workspace (labeled with numbers SITE##-####) is always associated with a main study and will have the most current reviewed information for that site.
- Continuing Review, Modification/Update, and Reportable New Information workspaces (webpages) are used actively during the review of that submission only. Once a determination is made (or the submission is discarded), Continuing Review, Modification/Update and Reportable New Information workspaces are used for reference only and should not be the go-to location for study information.

Workspace Regions
Image displays the Initial/Main study workspace for reference.