You can request that the Harvard IRB rely on the review of another IRB (referred to as External IRB or ceded review). This type of submission can be appropriate when a study is conducted at two or more different institutions, and instead of getting a separate IRB approval from each institution, an agreement is made for one institution to conduct the review and the other institution(s) to rely on that IRB’s review.
This submission type is only appropriate if you are requesting that Harvard rely on the review of another IRB. Once submitted, the Harvard IRB will determine if a reliance agreement is possible or if Harvard IRB review is required. To instead have the Harvard IRB conduct the review of the study see What to Include in a Study SmartForm.
The simplest approach is to follow the forms in order, answering the questions and clicking Continue to save your information and move to the next form. When you reach the end of the series of forms, click the Save & Exit button.
What to Include in the Initial Request for External IRB Review
To create a request for External IRB review see Creating a New Study. The SmartForm is the series of web-pages (or forms) where you can input specific information about a study. The following section describes what information should go into each space of the SmartForm.
Important! Completing elements of the Study/Site SmartForms or clicking “Save & Exit” do not send this request for review. The PI must visit the associated Site workspace and click Submit (marked with a red arrow in the submission workspace) for the request to proceed on for the next state of review.
SmartForm Section: Basic Study Information (External IRB)
1. Title of study. Enter the complete study title. Please avoid use of quotation marks in the study title.
2. Short title. The short title identifies the study throughout the system, such as in your inbox and in an IRB reviewer's list of submissions to review. This should be 80 characters or less, and should not contain quotation marks. If left blank, the system will automatically pull in the first 80 characters of the (long) study title.
3. Brief description or abstract. Enter a brief description of the study or the study abstract. This should be 225 words or less.
4. What kind of study is this?
The options selected here will show additional features in other sections of the system.
Select Multi-Site or Collaborative to allow for the entry/addition of information about study sites and if the project may involve:
• More than one site conducting the study (completely or in part), or
• Collaborators with primary affiliation with other institutions, or
• If it is unclear if there will be additional sites.
Select Single-site Study if the project only involves work at Harvard, by Harvard affiliated researchers. Note that a request for External IRB review will rarely be a Single-site type project.
Note: The remainder of this instruction in this section shows the longest possible form which displays when “Multi-Site or Collaborative” is selected here. However, expect the following pages to display based on the selection made:
|
Multi-Site or Collaborative |
Single-site Study |
|
|
4. Will an external IRB act as the IRB of record for this study? Select Yes. When you selected Yes, the attachment space on the Basic Information SmartForm will disappear (it is not needed when requesting External IRB review).
Important! Once this selection is made and then saved, it cannot be changed. If the incorrect selection is made or if this must be changed after initial review, the submission will need to be recreated.
5. Lead Principal Investigator. (only displays if question 4 is marked “Multi-site or Collaborative” AND question 5 is marked “Yes”). The options are limited to individuals with a Harvard ID/HarvardKey. If the Lead Principal Investigator name cannot be found on the list, leave this space blank. For details on who may be listed in this space, please visit your IRB website or Investigator Manual.
6. Local Principal Investigator. Enter the name of the Harvard Principal Investigator. The options are limited to individuals with a Harvard ID/HarvardKey. For details on who may be listed in this space, please visit your IRB website or Investigator Manual.
This field will default to the name of the person completing the form. It may be changed by selecting clear and typing the name in the space provided (a list will appear with names from which to select), or you can click the select option to search the list of names. Use the “%” symbol to replace portions of a name which are unclear or possibly not spelled correctly. This is called a “wildcard” character.
5. Does the principal investigator have a financial interest related to this research? Indicate if the Principal Investigator has a financial interest. For more information regarding what constitutes a financial interest, please visit your IRB website. If an individual has a financial interest, complete and attach “FORM: Financial Interest Disclosure Form” to the Local Site Documents page of the associated Site SmartForm.
6. Study's department. Indicate the name of the Principal Investigator or Faculty Sponsor’s department for this study.
Type the department name in the space provided (a list will appear with options from which to select), or you can click the select option to search the list of available departments and schools. Use the “%” symbol to replace portions of a word of which may be unclear or possibly not spelled correctly. This is called a “wildcard” character.
If the department name does not appear on the list provided, contact estrhelp@harvard.edu for guidance on which department Once chosen, saved and submitted, the department cannot be changed during review. To change the department after a study is submitted (but before the first determination by the IRB):
1. Complete the Withdraw activity on the study in review
2. Click “Edit Study” to change the name of the department on the SmartForm
3. Click “Save” then “Exit” on the SmartForm menu
4. The PI must complete the Submit activity on the study to send it back to the IRB
SmartForm Section: Basic Local Site Information (External IRB)
Page only displays if this is a request for Harvard to rely on the review of another IRB and this is a multi-site or collaborative project.
Brief description of activities this site will perform: Provide information about activities taking place at Harvard or by Harvard researchers.
SmartForm Section: External IRB
On the following page you will need to enter information about the External IRB that will serve as the IRB of Record for the study.
1. External IRB: Click Select to indicate which institution will serve as the IRB of Record. If the reviewing institution is not listed, select Other.
2. External study ID: If available, the study number given by the External IRB.
3. Specify the reason the study should be reviewed by an external IRB: Write in the rationale for the request for Harvard to rely on another institution.
SmartForm Section: Funding Sources (External IRB)
On this page include any pending/awarded funding sources or financial support for this study, with the IRB of record. Leave questions 1 and 2 blank to indicate that there is no non-Harvard funding for this study. You will have an opportunity later in the forms to indicate funding sources specific to the Harvard site. Reminder: If the funding status changes following IRB determination, submit a modification to this study.
1. List any grant proposal that has been submitted to the sponsored programs office (SPA or OSP), any proposal that was created in GMAS, or any federal or other sponsored funding for the study. Find and add all funding sources listed in the Harvard Grants Management Application Suite (GMAS) that are associated with this study.
To search for funding in the space provided, begin typing the grant project ID, the grant PI full name (first and last name), or the GMAS fund number and a list will appear with options from which to select.
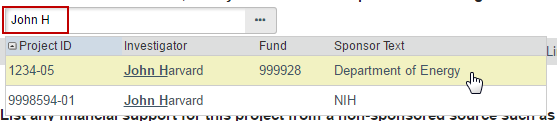
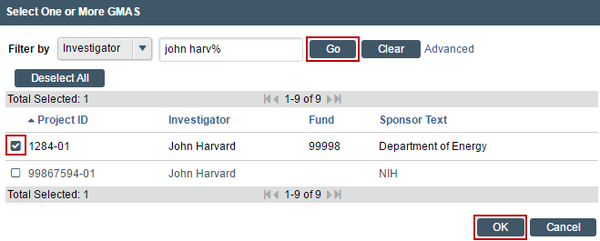
If additional search options are needed, can click the ellipses […] button option to search the list of available funding sources. Use the “%” symbol to replace portions of a word of which may be unclear or possibly not spelled correctly. Click “Go” and select the correct funding source. Click “OK” on the pop up.
2. List any financial support for this project from a non-sponsored source such as a department, gift or Harvard program. List all other funding sources.
SmartForm Section: Additional Local Funding Sources (External IRB)
Page only displays if this is a request for Harvard to rely on the review of another IRB and this is a multi-site or collaborative project. On this page include only funding that will be managed through this site that is also not included with the main study SmartForm review.
Identify any additional organization supplying funding for the local site. List all other funding sources. Attach a complete copy of the funding application or agreement for these listed sources to the Local Site Documents page of the SmartForm, via the document attachment prompt, when applicable, and remove or black-out any salary information.
SmartForm Section: (Local) Study Team Members (External IRB)
The Study Team Members page of the SmartForm should include the name of individuals that a) have contact with human subjects, b) have access to data that is identifiable; or c) are responsible for the design, conduct, or reporting of the research. Take care to follow the instructions that display on the SmartForm page.
1. List study team members with an HUID. Click “Add” to include all team members who have an HUID.
Individuals can be selected from a list of names. Begin typing a piece of the person’s name in the space provided on the pop up, or you can click the “add” option to search the list of available names. Use the “%” symbol to replace portions of a name of which may be unclear or possibly not spelled correctly. This is called a “wildcard” character. Use the email address column for search or as a guide to selecting the correct person. If the correct person cannot be found via name or email, click the ellipses […]. Choose Filter by: UserID to search by HUID
If you would like a person on the study team to access ESTR, and his/her name does not appear in the search results; this individual must obtain an HUID. Please visit the ESTR support site Job Aids page for detailed instructions on how to obtain an HUID for use with ESTR.
Each of the individuals listed must complete human research training unless you clearly indicate that the individual is listed simply for access to this study record (in the Study Team Member details and via “Add comment” on the submission workspace).
Indicate whether each listed individual has a financial conflict of interest relating to the Human Research. For more information about what constitutes a conflict, please visit your IRB website. If an individual has a financial interest, complete “FORM: Financial Interest Disclosure Form” and attach it to item 2 on this page of the SmartForm.
2. Attach relevant study team documents. If an individual’s name does not appear in the search results available in question 1, attach a completed “FORM: Non-Harvard Study Personnel Form” This form should only list individuals, who were not able to be listed under item 1 in the Study Team Members section of the SmartForm. Each of the individuals listed must complete human research training. Individuals only listed on this form will not have access to ESTR.
Indicate whether each listed individual has a financial conflict of interest relating to the Human Research. For more information about what constitutes a conflict, please visit your IRB website. If an individual has a financial interest, complete “FORM: Financial Interest Disclosure Form” and attach it here.
TIPS
> Do not list the PI on this page.
> For studies with a non-faculty Principal Investigator and where the IRB requires (as applicable, University Area only), list the Faculty Sponsor on this page.
> Each of the individuals named as study team members must complete human subjects training (refer to your IRB website to learn about training requirements).
SmartForm Section: Study Scope (External IRB)
1. Does the study involve the use of a drug in one or more persons other than use of an approved drug in the course of medical practice? Indicate if drugs, biologics, foods or dietary supplements are used in the Human Research.
SmartForm Sub-Section: Drugs
Identify all drugs, biologics, foods and dietary supplements (approved and unapproved) being used in the Human Research. For each, indicate whether it has an IND number and for those that do, ensure that the application includes one of the following: (a) sponsor protocol with the IND number; (b) communication from the sponsor with the IND number; or (c) communication from the FDA with the IND number. Additionally, attach the following items:
- Investigator Brochure for each investigational drug involved in the Human Research.
- Current Package Insert. Submit for each marketed drug involved in the Human Research.
- Validation of IND#, e.g. FDA Approval letter or Sponsor Protocol
2. Does the study involve the use of a DEVICE in one or more persons that evaluates the safety or effectiveness of that device, or data regarding the use of a device on human specimens? Indicate if a device is used in the Human Research.
SmartForm Sub-Section: Devices
Identify all devices being evaluated for safety or effectiveness or as a comparator (approved and unapproved). For each, indicate whether it has an IDE number and for those that do, ensure that the application includes one of the following: (a) sponsor protocol with the IDE number; (b) communication from the sponsor with the IDE number; or (c) communication from the FDA with the IDE number. Indicate whether the device is being submitted under the “Abbreviated IDE requirements” in 21 CFR 812.2(b). Additionally, attach the following items:
- Current Product Information, e.g., Device Manual. Submit for each investigational device involved in the Human Research.
- Validation of IND# or IDE#, e.g. FDA Approval letter or Sponsor Protocol
SmartForm Section: Research Location
Identify other research locations where the Harvard investigator will conduct or oversee the research. Indicate details as prompted on the research location details. For each location, the country is a required selection. If the location country is not available select the option “[country not listed]”.
Upload information for each location to the Local Site Documents page of the SmartForm, via the last document attachment prompt, when applicable.
SmartForm Section: Local Site Documents (External IRB)
Page only displays if this is a request for Harvard to rely on the review of another IRB and this is a multi-site or collaborative project.
Important! This page of the SmartForm is intended to capture only documents that (1) have not been included in other sections of the SmartForm AND will be used across all sites involved in the project.
1. Consent, Assent, Permission, and HIPAA Authorization Forms (English versions): Use the templates appropriate to your IRB to create and attach the following items. Visit your IRB website for additional instructions regarding how to create consent materials:
- Consent, Assent Forms, or Scripts including HIPAA Authorization Forms (as applicable). Consent/assent documents must include version date and/or version number. If any consent materials will be translated, attach the foreign language versions of materials to the last section of this page of the SmartForm.
- DHHS-approved sample consent document. Consent documents must include version date and/or version number.
If any of materials will be translated, attach the foreign language versions of materials to the last section on this page of the SmartForm.
2. Recruitment Materials (English versions): Each recruitment document, script, flyer, advertisement, etc. must include version date and/or version number. Guidance on what is appropriate to include and exclude within an advertisement can be found in “WORKSHEET: Advertisements” and/or “WORKSHEET: Payments.” Worksheets are available for reference on your IRB website or in the Library section of ESTR (linked at the top of the screen under the “IRB” heading). Advertising material must include version date and/or version number.
If any of materials will be translated, attach the foreign language versions of materials to the next section on this page of the SmartForm.
Attach other supporting files, naming each file as it should appear in the approval letter: Also, be sure to name the file itself in a manner that identifies it as associated with your study. The suggested best practice for file names: StudyNumber_DocumentName_VersionNumber
When uploading documents to this page, you are prompted to select the appropriate attachment category. Select the correct category when uploading the first version. Once chosen and saved, the attachment category cannot be changed. To change a category, a document must be removed and then re-uploaded with the new category.
Use the “other” category to attach non-document type attachments which must be part of the review (such as compressed/zip, video, audio, or htm file formats).
Items to attach to this section:
- Ancillary Approvals/Permissions. Submit approval letters or permissions from any additional office or organization reviewing this project (for example, a letter of support). If unavailable at the time of submission, plan to submit a copy to the IRB prior to implementing any study procedures.
- Debriefing Materials. If any of these materials will be translated, also submit a Translation Attestation Form.
- Research Location Information. If question 1 in the SmartForm Section: Study Scope indicated that there are research locations where the research will be conducted submit relevant approvals (such as a Community Advisory Board approval) or other information from each research location identified in the SmartForm. If unavailable at the time of submission, plan to submit a copy to the IRB prior to beginning any human subjects research at the location(s).
- Study Instruments/Tools. Including all data collection instruments, questionnaires, surveys, focus group discussion guides, or interview guides. Do not include Case Report Forms. Study documents must include version date and/or version number.
SmartForm Section: Local Site Documents (External IRB)
This page of the SmartForm is intended to capture documents that will be used (only) by the Harvard researchers. Ensure that the document versions attached here are relevant to for the specific site under review.
Attach other supporting files, naming each file as it should appear in the approval letter: Also, be sure to name the file itself in a manner that identifies it as associated with your study. The suggested best practice for file names: StudyNumber_DocumentName_VersionNumber
When uploading documents to this page, you are prompted to select the appropriate attachment category. Select the correct category when uploading the first version. Once chosen and saved, the attachment category cannot be changed. To change a category, a document must be removed and then re-uploaded with the new category.
Use the “other” category to attach non-document type attachments which must be part of the review (such as compressed/zip, video, audio, or htm file formats).
Items to attach to this page:
- Consent, Assent Forms, or Scripts including HIPAA Authorization Forms
- Recruitment Materials
- Study Instruments/Tools
- IRB/SmartIRB Cede Request or IRB Authorization Agreement (IAA). When multiple institutions are engaged in the Human Research, submit this form allowing an investigator to designate Harvard as the Reviewing Institution (responsible for IRB review). To request such an agreement with a SmartIRB participating institution, visit this link.
- Data use agreements or other Agreements. Submit copies of any documents authorizing the use of data, or other contracts or agreements associated with the study.
- Financial Interest Disclosure Form. Submit this form if the Principal Investigator self-identifies a financial conflict of interest related to the research on the SmartForm: Basic Information page. Forms for any other team members which self-identify a financial conflict of interest should be attached to the SmartForm: Study Team Members page.
- Funding Source Attachments. If not associated with GMAS and as applicable, submit a complete copy of the grant applications, subcontract, and/or any funding agreements regardless of funding source.
Upon clicking continue from this page, the browser will direct to the last page of the SmartForm where you will have the opportunity to select “Save & Exit” to return to the submission workspace. The PI must click Submit (marked with a red arrow in the submission workspace) for the submission to proceed on to the IRB for review.
When Harvard relies on the review of another IRB, the submission will be accepted into an Active or External IRB state (status).
Continuing Review or Closure of a project reviewed by an External IRB
To provide a status update (for continuing review or closure) of the Harvard site, the Harvard Principal Investigator must visit the associated site workspace to complete the Report Continuing Review Data activity (on the left side of the site workspace). Within the activity, provide status information and relevant attachments. To request closure, indicate the request in the text box provided on the activity. Note that only the Harvard Principal Investigator (named on the Harvard Site SmartForm) will see/have access to the Report Continuing Review Data activity.
Modification to a project reviewed by an External IRB:
Important! External IRB submissions completed prior to October 2015 cannot be edited via modification. Contact the IRB office to determine if an update is required. If one is required, the submission will need to be recreated under new requirements and processes.
For multi-site or collaborative projects that are in the Active state, study revisions and updates that are reviewed by the IRB of record can be recorded by either:
-
Updating Study Details to provide an update to Harvard about general status or study content and provide a copy of the approval letter from the IRB of record.
OR - Creating Site Modification to provide information about revisions to Harvard-site information or activities and provide a copy of the approval letter from the IRB of record.
For single site projects that are in the External IRB state, study revisions and updates that are reviewed by the IRB of record can be recorded by Updating Study Details to provide an update to Harvard about general status or study content and provide a copy of the approval letter from the IRB of record.
Update Study Details
- Log in to irb.harvard.edu
- Navigate to the External IRB’s associated site workspace (see Accessing a Submission for more information on finding the External IRB record, and clicking the site link).
- Click the “Update Study Details” button on the submission workspace, a SmartForm will open. Based on the selection, this option will only display the External IRB, Study Funding Sources, Study Scope, and Study-Related Documents pages of the SmartForm to edit.
- Provide a brief description of the contents of the updates.
- By selecting ‘Continue’ the system take you to a copy of the Site SmartForm. Refer to the What to Include in the Initial Request for External IRB, Site SmartForm section for additional instructions for what is required for each element of this SmartForm.
- Follow the remaining steps to Edit a Submission or Save & Exit when edits are complete.
The PI or PI Proxy must click Finalize Updates (on the left side of the submission workspace) for the Harvard IRB to be notified that an update has occurred.
Create Site Modification
- Log in to irb.harvard.edu
- Navigate to the External IRB’s associated site workspace (see Accessing a Submission for more information on finding the External IRB record, and clicking the site link).
-
Click the “Modify Site or “Create Site Modification” button on the site workspace, a SmartForm will open. For Modification scope, if “other parts of the site” is chosen, this option will allow for edits to only the Additional Local Funding Sources, Local Research Locations and Local Site Documents pages of the SmartForm to edit. If “team members” is chosen, this option will allow for edits to the Local Study Team Members page of the SmartForm. One or both options may be selected when indicating the scope of the modification on the SmartForm.
Note: If the “Modify Site” or "Create Site Modification" button is not visible, be sure that: (1) a primary contact is assigned to the site and (2) as appropriate, an IRB coordinator is assigned to the site. One or both of these additions will update the workspace for the “Modify Site” or “Create Site Modification” button to display.
- Provide a brief description of the contents of the modification, along with the scope.
- By selecting ‘Continue’ the system take you to a copy of the Site SmartForm. Refer to the What to Include in the Initial Request for External IRB, Site SmartForm section for additional instructions for what is required for each element of this SmartForm.
- Follow the remaining steps to Edit a Submission or Save & Exit when edits are complete.
- The PI or PI Proxy must click Submit (marked with a red arrow in the submission workspace) for the submission to proceed on to the IRB for review.
Finding External IRB Review Projects by Report
To generate a report listing all studies where you serve as the Harvard Principal Investigator (PI) where Harvard has ceded IRB review to another institution:
- Log in to irb.harvard.edu
- Click IRB in the top navigator.
- Click Reports in the IRB sub-menu. The Standard Reports tab is the default view.
- Under Standard Reports, locate the All External Studies Report on the list and click the hyperlink.
- A report will appear listing all submissions where you are listed as the Harvard PI on a project where Harvard has ceded IRB review to another institution.
- Report results can be exported to Excel from the results pop up window (see below for more information).
For more general information, reference the Generating Standard Reports guidance.